 
weak acids and strong bases are soluble. 
strong acids and strong bases are soluble. 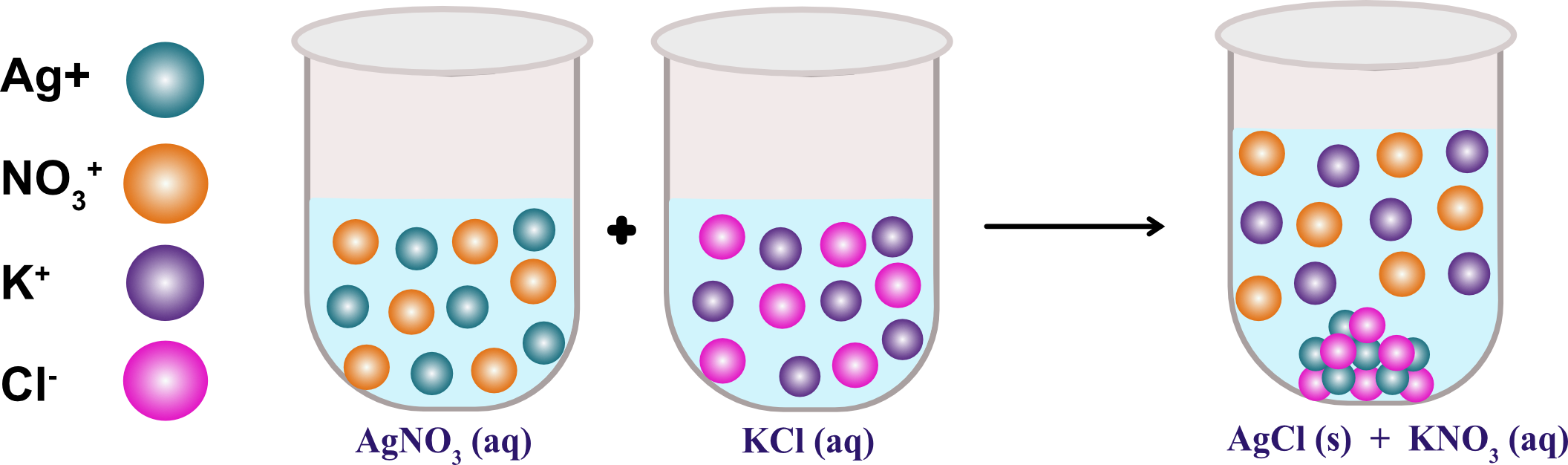
In the equation, NaCl and KI are the reactants, and NaI and KCl are the products. NaCl (aq) + KI (aq) → NaI (aq) + KCl (aq) The following equation shows a double replacement reaction between sodium chloride (NaCl) and potassium iodide (KI). In this type of reaction, the positive ions of one compound exchange places with the positive ions of the other compound. When two compounds interact to produce a new compound and two molecules of water, the process called a double replacement reaction. Precipitation and Double Replacement Reactions
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
