 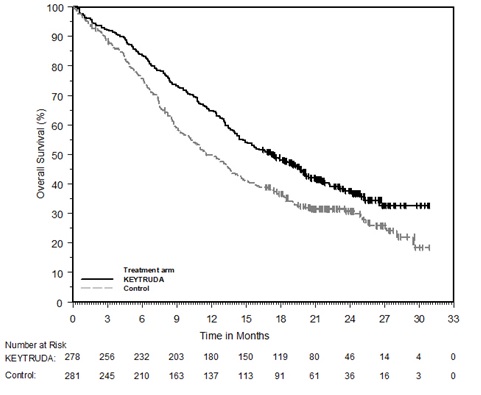
These data indicate a durable effect at 1 year following completion of treatment.Ī population of patients with PD-L1 expression of at least 50% also demonstrated benefit with IO in the IMpower110 study (NCT02409342), in which investigators evaluated the efficacy of atezolizumab (Tecentriq) versus chemotherapy. Of note, the pembrolizumab regimen included up to 2 years of IO and reported a 31% survival rate for those who received IO at 3 years. The study confirmed the benefit of pembrolizumab compared with chemotherapy in the population of patients with PD-L1 TPS of at least 50%, also evaluated by IHC 22C3. Eligible patients were adults (≥18 years). 2 The KEYNOTE-042 (NCT02220894) 3 open-label, phase 3 study was conducted in 213 medical centers in 32 countries. Investigators reported a median over-all survival (OS) of 30.0 months (95% CI, 18.3–not reached ) in the pembrolizumab arm and 14.2 months (95% CI, 9.8-19.0) in the chemotherapy arm with a hazard ratio of 0.63 (95% CI, 0.47-0.86) in patients with a PD-L1 tumor proportion score (TPS) of at least 50% by immunohistochemistry (IHC) 22C3 pharmDX assay. In KEYNOTE-024 (NCT02142738), 1 pembrolizumab (Keytruda) was com-pared with platinum-based chemotherapy. Several studies have evaluated the role of immunotherapy as monotherapy in patients with PD-L1 expression of at least 50%. PD-L1–High Metastatic NSCLC: Immunotherapy Alone Data from completed studies evaluating IO alone or in combination with chemotherapy show that a breakdown of populations by PD-L1 status and additional considerations such as toxicity profiles should guide treatment decisions. Recent studies have demonstrated a role for immuno-oncology (IO) agents in the first line, but the added benefit of combining them with chemotherapy (chemo IO) is not clear in all populations. Despite these advances, the best treatment options for patients with nondriver met-astatic NSCLC remain unclear. Progress made in the area of targeted therapies has pushed treatment with small-molecule inhibitors to the front-line setting for patients with aberrations in EGFR, ALK, ROS1, BRAF, MET, and RET. The therapeutic landscape for first-line treatment of metastatic non–small cell lung cancer (NSCLC) contains many options. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
